|
(7,9,10) In contrast, serine proteases show a catalytic triad motif, consisting of an aspartate, a histidine, and a serine, of which only the aspartate is negatively charged, while the histidine and serine are ionized intermediately during catalysis. In cysteine proteases on the other hand, a cysteine and a histidine constitute the active site, which form an ion pair, i.e., the cysteine is in its thiolate form, while the imidazole side chain of the histidine is protonated and therefore positively charged. (3,8,9) For this purpose, it is imperative that the dyad is in a monoprotonated state when the protease is active.  
The catalytic center of aspartate proteases consists of an aspartic dyad, of which one aspartate acts as a base and the other one as an acid during catalysis. The major distinctions between these three families in terms of catalysis can be found in their active site architecture. While aspartic proteases are active in the acidic range, cysteine proteases cover the mild acidic to neutral range and finally serine proteases are mostly found active at neutral to slightly alkaline conditions. 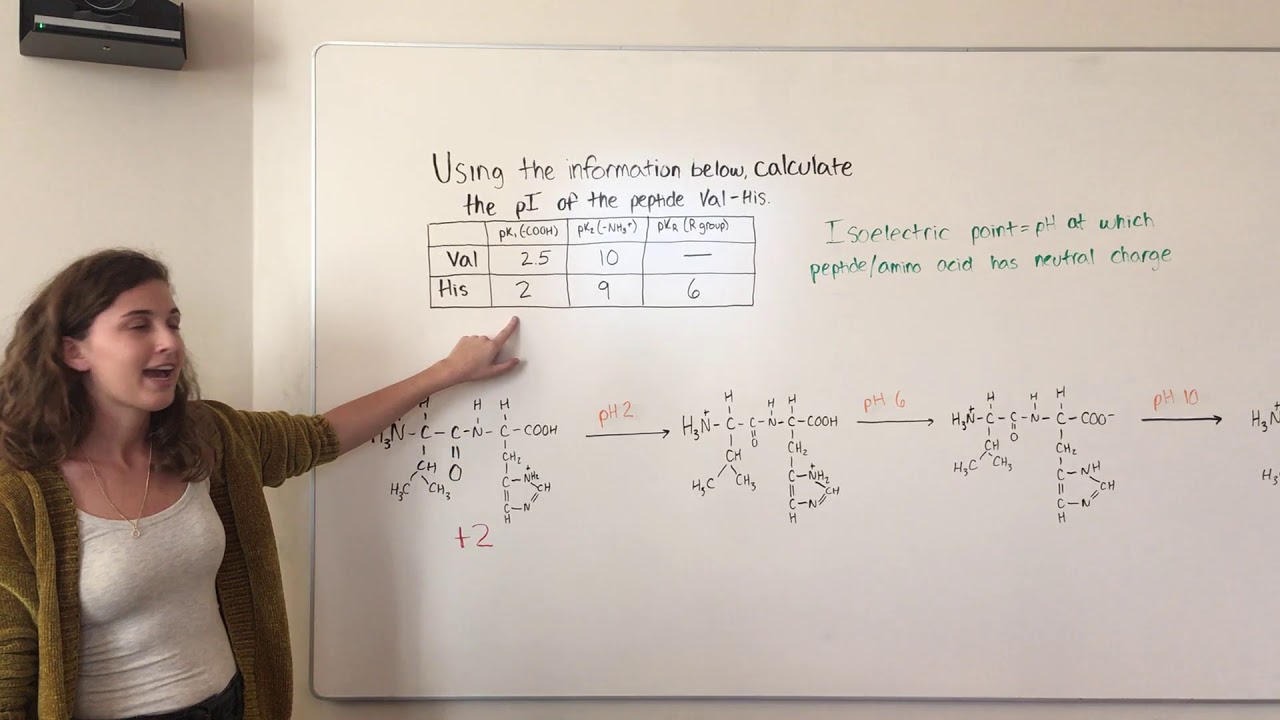
(5,6) Taken together, these three families cover a broad pH range in terms of activity. (3,4) An overview of the various activity profiles of aspartic, cysteine, and serine proteases is shown in Figure 1. For example, digestive proteases in the stomach at a pH of 2.0 have to catalyze the same reaction as proteases of the blood coagulation cascade at a pH of 7.4 and proteases in the gut at basic conditions. (2) Consequently, the physiological environments where proteases need to operate are very diverse as well, including vastly different ranges of acidity. Processes from signaling cascades over digestion to programmed cell death are based on proteolytic processing. (1) The function of the proteases is manifold. Indeed, 2–3% of all human genes code for proteases or protease inhibitors. Proteases catalyze the cleavage of peptide bonds, a ubiquitous reaction in the whole biosphere. Consequently, with this work, we provide a concise set of active site p K a values of aspartic and serine proteases, which could serve as reference for future theoretical as well as experimental studies. While we find convincing agreement with experimental data for serine and aspartic proteases, we observe clear deficiencies in the description of the titration behavior of cysteines within the constant pH MD framework and highlight opportunities for improvement. These shifts are caused by a distinct network of electrostatic interactions characteristic for each protease family. We find clear differences of active site p K a values within the respective families, which closely match the experimentally determined pH preferences of the respective proteases. We capture characteristic p K a shifts of the active site residues, which dictate the experimentally determined activity profiles of the respective protease family. In this study, we use single pH constant pH MD simulations as a fast and robust tool to estimate the active site p K a values of a set of aspartic, cysteine, and serine proteases. However, experimental p K a data are scarce, since the systems’ size and complexity make determination of these p K a values inherently difficult. Knowledge of the titration behavior of these residues is crucial for the development of drugs targeting the active site residues. The protonation states of titratable residues in the active site react to changes in the pH value, according to their p K a, and thereby determine the functionality of the enzyme. 
Enzymatic function and activity of proteases is closely controlled by the pH value.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
