|
This radioactive decay result can also be derived in a very simple way of learning chemistry or physics. 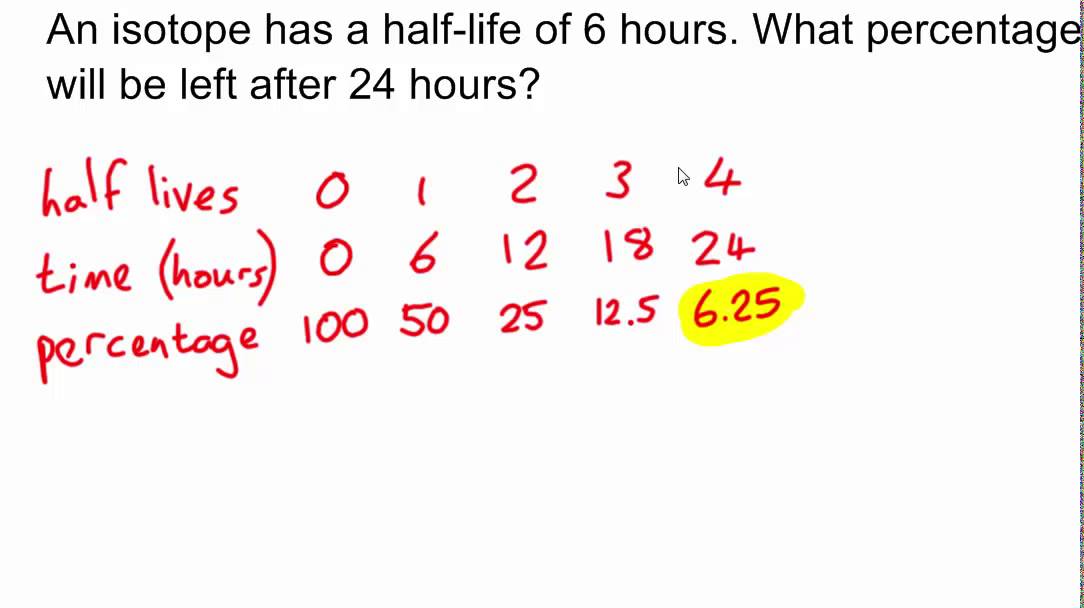
The mean life of radio-element is reciprocal of its radioactive decay constant in a nuclear reaction. The total number of radioactive atoms (N 0) is composed of many small numbers of atoms like dN 1, dN 2, dN 3, etc, each with its own life span t 1, t 2, t 3, etc. This explains the gradual radioactive decay of the radioisotopes instead of the decay of all the atoms at the same time. It is the length of time a radioelement can live before it disintegrates that may have values from zero to infinity. The mean life period of an atom of the radioactive element tells us the average span of time after which the atom will disintegrate. Hence, after complete n half-lives, the amount of radioactive element reaming (1/2) n.After 8 hours it decomposes 75% and reaming 25% and the process continued.After four hours, it decomposes 50% and the remaining 50%.If the radioactivity of an element is 100% and the half-life period of this element is 4 hours. The half-life of polonium-213 isotope = 4.2 × 10 −6 sec and bismuth-209 isotope = 3 × 10 7 years. Hence the half-life is independent of the amount of the radio element present at a given time. This equation provides the relation between half-life and radioactive decay constants. Half-life is a characteristic property of radioactive elements. This period of decay is called the half-life of radioactive material or substances. What is half-life period?Īfter a certain period of time, the value of (N 0/N ) becomes one-half of the radioactive elements. The negative sign shows that N decreases with time. Therefore, the radioactive decay constant represents the fraction of radioactive atoms that disintegrates in a unit time. The quantity dN/N represents the fraction of the total number of atoms that disintegrate in time dt. The radioactive decay formula is given below the picture, 
If N 0 number of the atoms present at the time t = 0 and N number of atoms present after the time t. Therefore, dN/dt = − kN, where N = number of the atoms of the disintegrating radioactive element, dt = time over which the decay is measured, and k = radioactive decay constant. The rate of radioactive decay formula depends only on the single power of the radioactivity. It is used mainly in radiocarbon dating and the determination of the age of matter or materials. Radioactive decay is one important natural phenomenon obeying the first-order chemical kinetics. In chemistry, the half-life is the period of time when half of the radioactive substances undergo disintegration. Soddy also coined the term radioactive isotopes which occupy the same place in the periodic table. 
According to them, atom of radioactive elements undergo spontaneous disintegration to form a new atom of elements. Rutherford and soddy 1902 proposed the law of radioactive decay. Therefore, radioactive decay is simply determined by counting the number of alpha, beta, and gamma radiation in a given time. Radioactive decay is the spontaneous disintegration or emission of atomic particles like alpha, beta, gamma from the nuclei of radioactive substances in the form of nuclear energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
